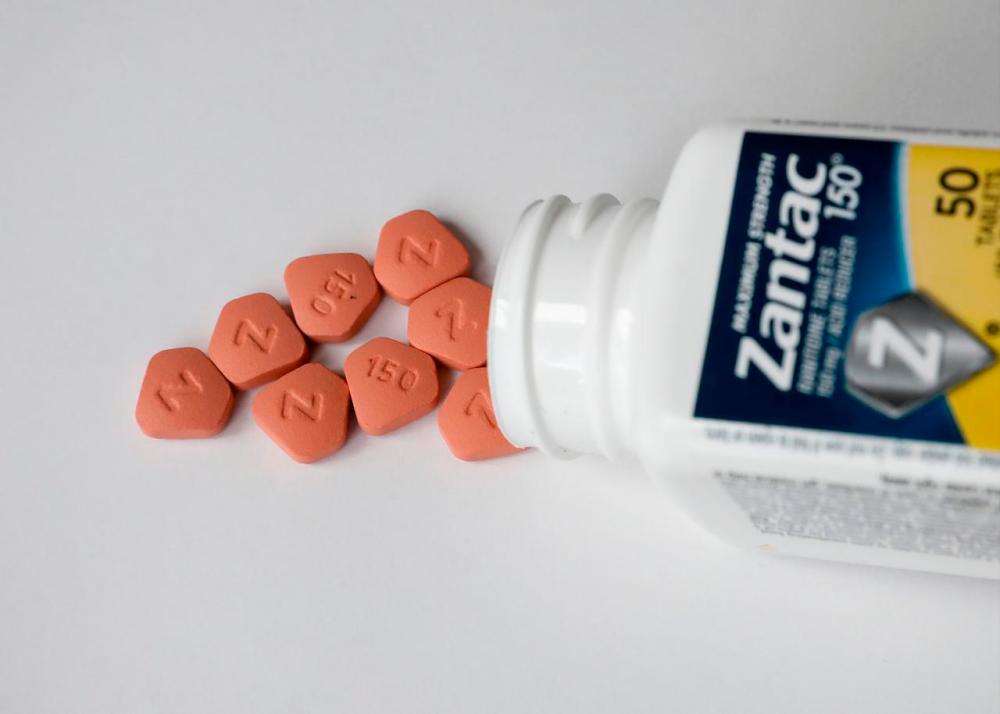
PETALING JAYA: Pharmaceutical companies in Malaysia have until Nov 7 to test whether the gastric pills ranitidine contains the impurity N-Nitrosodimethylamine (NDMA) with the National Pharmaceutical Regulatory Agency (NPRA).
The NDMA has identified as a potential carcinogen.
The Health Ministry said in a statement dated Sept 20, that all companies involved have been instructed to stop distribution and supply of these products until investigation and evaluation by the NPRA is completed.
This follows news that Singapore, the US and Canada has recalled the product of ranitidine following discovery that some of its products contain NDMA.
Ranitidine is a drug that is used to treat heartburn and stomach ulcer, but NDMA can increase the chance of one to get cancer on a long term basis.
A circular dated Oct 25 and sighted by theSun also found that Pharmaniaga Berhad had voluntarily recalled their ranitidine products from hospitals and pharmacies.